New York, USA, April 07, 2026 (GLOBE NEWSWIRE) -- Severe Asthma Clinical Trial Pipeline Appears Robust With 30+ Key Pharma Companies Actively Working in the Therapeutics Segment | DelveInsight | DelveInsight
The severe asthma clinical trial analysis report delivers important insights into ongoing research of 35+ pipeline severe asthma drugs, clinical strategies, upcoming therapeutics, and commercial analysis.
DelveInsight’s Severe Asthma Pipeline Insight 2026 report provides comprehensive global coverage of pipeline therapies for severe asthma across various stages of clinical development. The report offers an in-depth analysis of key trends, emerging therapies, and competitive landscape dynamics, highlighting the strategies of major pharmaceutical companies to advance the pipeline and capitalize on future growth opportunities. In addition, it includes critical insights into clinical trial benchmarking, partnering and licensing activities, and regulatory pathways involving the FDA and EMA, enabling stakeholders to make informed decisions and optimize development strategies within the severe asthma domain.
Severe Asthma Clinical Trial Analysis Summary
- DelveInsight’s severe asthma pipeline report depicts a robust space with 30+ active players working to develop 35+ pipeline severe asthma drugs.
- Key severe asthma companies such as CSPC ZhongQi Pharmaceutical Technology, Oneness Biotech, Biosion, Suzhou Connect Biopharmaceuticals, Upstream Bio, Teva Branded Pharmaceutical Industries, Lanier Biotherapeutics, Nanjing RegeneCore Biotech Co., Ltd., AstraZeneca, Generate Biomedicines, Incyte Corporation, Shanghai Hengrui Pharmaceutical Co., Ltd., and others are evaluating new severe asthma drugs to improve the treatment landscape.
- Promising pipeline severe asthma therapies, such as CM326, FB704A, BSI-045B, CBP-201, Verekitug, TEV-56248, LNR 125.38, RC1416, Tezepelumab, GB-0895, Povorcitinib, CM310, SHR-1905, and others, are in different phases of severe asthma clinical trials.
- Approximately 15+ severe asthma drugs are in the mid stage of development, whereas 8+ drugs are in the early stages of development.
- Notable MoAs in severe asthma clinical trials include Antioxidants, Apoptosis inhibitors, Dopamine receptor agonists, Free radical scavengers, Thymic stromal lymphopoietin inhibitors, Interleukin 6 inhibitors, Interleukin 4 receptor alpha subunit antagonists, CRLF2 protein antagonists, Beta 2 adrenergic receptor agonists, Steroid receptor agonists, Interleukin 4 receptor antagonists, Interleukin 5 inhibitors, and others.
Request a sample and discover the recent advances in severe asthma drugs @ https://www.delveinsight.com/sample-request/severe-asthma-pipeline-insight
What is Severe Asthma?
Severe asthma is a chronic respiratory condition in which asthma symptoms remain uncontrolled despite the regular use of high-dose inhaled corticosteroids and additional controller medications, or worsen when treatment is reduced. People with severe asthma experience frequent and intense symptoms such as breathlessness, wheezing, chest tightness, and coughing, often accompanied by recurrent exacerbations that may require oral steroids, emergency care, or hospitalization. The disease is characterized by persistent airway inflammation, bronchial hyperresponsiveness, and airway remodeling, leading to significant impairment in lung function and quality of life. Severe asthma is not a single disease entity but a heterogeneous condition with multiple underlying phenotypes and endotypes, necessitating individualized assessment and targeted treatment approaches.
Find out more about severe asthma drugs @ Severe Asthma Treatment
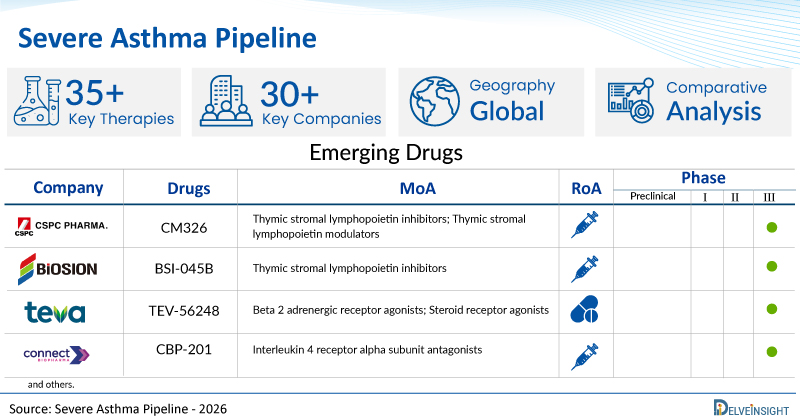
A snapshot of the Pipeline Severe Asthma Drugs mentioned in the report:
| Drugs | Company | Phase | MoA | RoA |
| CM326 | CSPC Pharmaceutical Group | III | Thymic stromal lymphopoietin inhibitors; Thymic stromal lymphopoietin modulators | Subcutaneous |
| BSI-045B | Biosion | III | Thymic stromal lymphopoietin inhibitors | Subcutaneous |
| TEV-56248 | Teva Branded Pharmaceutical Industries | III | Beta 2 adrenergic receptor agonists; Steroid receptor agonists | Oral inhalation powder |
| CBP-201 | Suzhou Connect Biopharmaceuticals | III | Interleukin 4 receptor alpha subunit antagonists | Subcutaneous |
| FB704A | Oneness Biotech | II | Interleukin 6 inhibitors | Intravenous |
| RC1416 | Nanjing RegeneCore Biotech Co., Ltd. | II | Interleukin 4 receptor antagonists, Interleukin 5 inhibitors | Subcutaneous |
| Verekitug | Upstream Biosciences | II | CRLF2 protein antagonists | Subcutaneous |
Learn more about the emerging severe asthma therapies @ Severe Asthma Clinical Trials
Stuti Mahajan, consulting manager at DelveInsight, said that the rise in severe asthma cases propels demand for advanced treatment options, spurring innovation and investment in pharmaceutical research and development.
Recent Developments in Severe Asthma Treatment Space
- In February 2026, Upstream Bio, Inc., a clinical-stage company developing treatments for inflammatory diseases, with an initial focus on severe respiratory disorders, announced positive top-line results from the Phase II VALIANT clinical trial evaluating the safety and efficacy of verekitug in adults with severe asthma.
- In February 2026, in the pooled analysis of KALOS and LOGOS, BREZTRI also demonstrated clinically meaningful reductions in the annualized rate of severe asthma exacerbations versus ICS/LABA medicines in patients with or without a recent asthma exacerbation.
- In February 2026, Generate Biomedicines’ lead asset, GB-0895, targets severe asthma and COPD, with Phase III enrollment expected to be completed by H2 2027.
- In January 2026, GSK announced that it had entered a multi-year strategic collaboration agreement with Helix, aiming to drive the development of precision medicines. The announcement comes on the heels of the FDA’s approval of GSK’s Exdensur for adolescents and adults with severe asthma characterized by an eosinophilic phenotype.
- In January 2026, A clinical trial to determine the best intravenous (IV) treatments for children with severe asthma will go ahead thanks to an almost $5 million Medical Research Future Fund grant. The clinical trial spearheaded by Professor Simon Craig, Adjunct Clinical Professor in the School of Clinical Sciences at Monash Health and paediatric emergency medicine physician at Monash Medical Centre, will provide clinicians with clear evidence to make more informed decisions, provide faster and more effective treatment, reduce unnecessary variation between hospitals, and ultimately improve outcomes for children with severe asthma.
- In December 2025, the FDA had approved GSK’s Exdensur (depemokimab-ulaa) for the treatment of severe asthma as an add-on maintenance therapy in adult and pediatric patients 12 years and older.
- In December 2025, Generate Biomedicines announced its plans to initiate two global Phase III clinical trials, SOLAIRIA-1 and SOLAIRIA-2, evaluating GB-0895 in approximately 1,600 adults and adolescents with severe asthma whose disease remains inadequately managed on current therapies. GB-0895 is an investigational, long-acting monoclonal antibody engineered with AI to target thymic stromal lymphopoietin (TSLP), a key driver of airway inflammation. These studies will evaluate the efficacy of GB-0895 in reducing clinically significant asthma exacerbations over 52 weeks, the primary objective for both trials.
Scope of the Severe Asthma Pipeline Report
- Coverage: Global
- Therapeutic Assessment By Product Type: Mono, Combination, Mono/Combination
- Therapeutic Assessment By Clinical Stages: Discovery, Pre-clinical, Phase I, Phase II, Phase III
- Therapeutics Assessment By Route of Administration: Oral, Intravenous, Subcutaneous, Parenteral, Topical
- Therapeutics Assessment By Molecule Type: Recombinant fusion proteins, Small molecule, Monoclonal antibody, Peptide, Polymer, Gene therapy
- Therapeutics Assessment By Mechanism of Action: Antioxidants, Apoptosis inhibitors, Dopamine receptor agonists, Free radical scavengers, Thymic stromal lymphopoietin inhibitors, Interleukin 6 inhibitors, Interleukin 4 receptor alpha subunit antagonists, CRLF2 protein antagonists, Beta 2 adrenergic receptor agonists, Steroid receptor agonists, Interleukin 4 receptor antagonists, Interleukin 5 inhibitors
- Key Severe Asthma Companies: CSPC ZhongQi Pharmaceutical Technology, Oneness Biotech, Biosion, Suzhou Connect Biopharmaceuticals, Upstream Bio, Teva Branded Pharmaceutical Industries, Lanier Biotherapeutics, Nanjing RegeneCore Biotech Co., Ltd., AstraZeneca, Generate Biomedicines, Incyte Corporation, Shanghai Hengrui Pharmaceutical Co., Ltd., and others
- Key Severe Asthma Pipeline Therapies: CM326, FB704A, BSI-045B, CBP-201, Verekitug, TEV-56248, LNR 125.38, RC1416, Tezepelumab, GB-0895, Povorcitinib, CM310, SHR-1905 and others
Dive deep into rich insights for new severe asthma treatments, visit @ Severe Asthma Drugs
Table of Contents
| 1. | Severe Asthma Pipeline Report Introduction |
| 2. | Severe Asthma Pipeline Report Executive Summary |
| 3. | Severe Asthma Pipeline: Overview |
| 4. | Analytical Perspective In-depth Commercial Assessment |
| 5. | Severe Asthma Clinical Trial Therapeutics |
| 6. | Severe Asthma Pipeline: Late-Stage Products (Pre-registration) |
| 7. | Severe Asthma Pipeline: Late-Stage Products (Phase III) |
| 8. | Severe Asthma Pipeline: Mid-Stage Products (Phase II) |
| 9. | Severe Asthma Pipeline: Early-Stage Products (Phase I) |
| 10. | Severe Asthma Pipeline Therapeutics Assessment |
| 11. | Inactive Products in the Severe Asthma Pipeline |
| 12. | Company-University Collaborations (Licensing/Partnering) Analysis |
| 13. | Key Companies |
| 14. | Key Products in the Severe Asthma Pipeline |
| 15. | Unmet Needs |
| 16. | Market Drivers and Barriers |
| 17. | Future Perspectives and Conclusion |
| 18. | Analyst Views |
| 19. | Appendix |
For further information on the severe asthma cure research, reach out @ Medication for Severe Asthma Treatment
Related Reports
Severe Asthma Epidemiology Forecast
Severe Asthma Epidemiology Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted severe asthma epidemiology in the 7MM, i.e., the United States, EU5 (Germany, Spain, Italy, France, and the United Kingdom), and Japan.
Severe Asthma Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key severe asthma companies, including AstraZeneca plc, GlaxoSmithKline plc, Sanofi S.A., Novartis AG, Roche Holding AG, Teva Pharmaceutical Industries Ltd., Merck & Co., Inc., Boehringer Ingelheim International GmbH, Regeneron Pharmaceuticals, Inc., Johnson & Johnson, Pfizer Inc., Bristol Myers Squibb, Eli Lilly and Company, Vertex Pharmaceuticals Incorporated, Amgen Inc., AbbVie Inc., Genentech, Inc., Gilead Sciences, Inc., Biogen Inc., CSL Limited, among others.
Asthma Market Insights, Epidemiology, and Market Forecast – 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology, as well as the market trends, market drivers, market barriers, and key asthma companies including GlaxoSmithKline, 4D Pharma plc, AstraZeneca, Suzhou Connect Biopharmaceuticals, Avillion, Pearl Therapeutics, ARS Pharmaceuticals, Sinomab, Avalo Therapeutics, Kymab, Sanofi, Cumberland Pharmaceuticals, Genentech, Inc., among others.
Asthma Clinical Trial Analysis
Asthma Pipeline Insight – 2026 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key asthma companies, including Mabpharm Limited, Avillion LLP, GlaxoSmithKline, Immunotek SL, AB Science, AstraZeneca, Inmunotek, Sterna Biologics, Verona Pharma, MediciNova, Foresee Pharmaceuticals, T-Balance Therapeutics, Teva Pharmaceutical Industries, Aldeyra Therapeutics, Trio Medicines, AstraZeneca, Cumberland Pharmaceuticals, Concentrx Pharmaceuticals, Tetherex Pharmaceuticals, SolAeroMed, Palobiofarma, Sanofi, Oneness Biotech, Novartis, Keymed Biosciences, Suzhou Connect Biopharmaceuticals, 4D Pharma Plc, Hoffman-La-Roche, Celltrion, Glenmark Pharmaceuticals, Archivel Farma, Siolta Therapeutics, Areteia Therapeutics, TFF Pharmaceuticals, Evelo Biosciences, Sunshine Guojian Pharmaceutical, Mabwell (Shanghai) Bioscience, Kinaset Therapeutics, Akari Therapeutics, KLUS Pharma, Upstream Bio, LEO Pharma, among others.
Chronic Obstructive Pulmonary Disease Clinical Trial Analysis
Chronic Obstructive Pulmonary Disease Pipeline Insight – 2026 report provides comprehensive insights about the pipeline landscape, pipeline drug profiles, including clinical and non-clinical stage products, and the key COPD companies, including Sanofi, Chiesi Farmaceutici S.p.A., United Therapeutics Corporation, Verona Pharma plc, Immunotek, Yungjin Pharm. Co., Ltd., Pulmotect, Inc., Tetherex Pharmaceutical, CSL Behring, AstraZeneca, Novartis, Genentech, Vertex Pharmaceuticals, EmeraMed, Afimmune, Mereo BioPharma, Synairgen, Adamis Pharmaceuticals, Quercegen Pharmaceuticals LLC, Regend Therapeutics, Meridigen Biotech Co., Ltd., Pulmatrix, Eisai, GlaxoSmithKline, EpiEndo Pharmaceuticals, 3SBio, OmniSpirant, Foresee Pharmaceuticals, Amgen, Organicell Regenerative Medicine, Arrowhead Pharmaceuticals, ProterixBio, RS BioTherapeutics, MitoRx, C4X Discovery, Respiratorius, ARK biosciences, Incannex, GNI Pharma, Celon pharma, Alveolus Bio, Kinaset therapeutics, Landos Biopharma, Parion Sciences, KeyMed Biosciences, Bioneer corporation, AlgiPharma, Palobiofarma, Dimerix Bioscience, Glenmark Pharmaceuticals, among others.
About DelveInsight
DelveInsight is a leading Business Consultant and Market Research firm focused exclusively on life sciences.

Contact Us Shruti Thakur info@delveinsight.com +14699457679 www.delveinsight.com
